
 June 17, 2010 - New policy adopted today, June 14th by the American Medical Association (AMA) recommends that electronic cigarettes (e-cigarettes) be classified as drug delivery devices, subject to the same FDA regulations as all other drug delivery devices. Additional policy adopted supports prohibiting the sale of e-cigarettes that are not FDA approved.
June 17, 2010 - New policy adopted today, June 14th by the American Medical Association (AMA) recommends that electronic cigarettes (e-cigarettes) be classified as drug delivery devices, subject to the same FDA regulations as all other drug delivery devices. Additional policy adopted supports prohibiting the sale of e-cigarettes that are not FDA approved.
The importation of e-cigarettes will be banned indefinitely as the result of a unanimous ruling by the U.S. Court of Appeals on February 3, 2010 which provides strong support for the FDA's position. (U.S. - federal appeals court, import of e-cigarettes on hold again..
The FDA had seized shipments of e-cigarettes beginning in 2008 as illegal drug-delivery devices. The FDA maintains that such devices are illegal because -- unlike other products which administer nicotine such as gums, patches, inhalers, and sprays -- e-cigarettes have not been submitted to the FDA with proof they are safe and effective.
AMA Board Member Edward L. Langston, M.D.: "Very little data exists on the safety of e-cigarettes, and the FDA has warned that they are potentially addicting and contain harmful toxins. Our new policies can help ensure that e-cigarettes are properly classified and regulated."
Click to enlarge..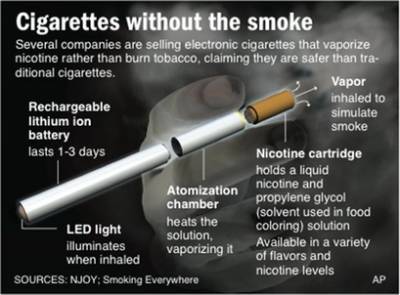 E-cigarettes are smokeless devices that deliver nicotine to the user. They consist of three integrated parts: the nicotine cartridge, the vaporizer and a lithium ion battery. The battery powers the cartridge and releases the nicotine by heating, rather than burning like a conventional cigarettes. They are available in fruit and candy flavors. Little independent research has been conducted into their ingredients and health impacts, but they are commercially promoted by vendors as a safe alternative to cigarettes.
E-cigarettes are smokeless devices that deliver nicotine to the user. They consist of three integrated parts: the nicotine cartridge, the vaporizer and a lithium ion battery. The battery powers the cartridge and releases the nicotine by heating, rather than burning like a conventional cigarettes. They are available in fruit and candy flavors. Little independent research has been conducted into their ingredients and health impacts, but they are commercially promoted by vendors as a safe alternative to cigarettes.
"Because e-cigarettes have not been thoroughly tested, one cannot conclude that they are less harmful or less dangerous than conventional cigarettes," said Dr. Langston. "The fact that they come in fruit and candy flavors gives them the potential to entice new nicotine users, especially teens."
The AMA's new policy is a result of a report looking at the current regulations and potential health impacts of e-cigarettes discussed this week at the AMA Annual Meeting in Chicago.
Electronic cigarettes - study finds not an effective nicotine delivery system..;
E-cigarettes need to establish efficacy and safety - FIRST..
We don't understand why these electronic cigarette devices continue to be sold in the United States while there is a ban on the sale of these imported products.
Reference: AMA: Electronic Cigarettes are Drug Delivery Devices and Should Be Subject to FDA Regulations, SOURCE American Medical Association, PR Newswire, 6/14/2010.
Bringing the World of Tobacco Control closer together..
US AMA policy - e-cigarettes FDA should treat as a drug delivery device..
Subscribe to:
Post Comments (Atom)


To Provide Public Awareness
Purpose
About Us
Contact Us
2008 HIGHLIGHTS
TOPIX PAPERS - 2008 & 2009..
Archive
-
▼
2010 (1530)
-
▼
06/13 - 06/20 (28)
- Bulgaria - NO decrease in cigarette excise tax..
- Bulgaria - tobacco growers again mass protests ov...
- Letter from US Senators (2) to tobacco companies..
- U.S. Senators warn tobacco companies not to engag...
- PMUSA store sign Marlboro 'New Look Same Famous F...
- PAPER: Main risk factors for stroke - hypertensi...
- FDA CTR - demanding that PM USA turn over resea...
- Plymouth, England - children could be banned from ...
- Reynolds American Inc. - cigarette ads in magazine...
- Western Australia - strict anti-smoking laws come...
- US AMA policy - e-cigarettes FDA should treat as ...
- Australia - Anne Jones, CEO of ASH chosen to recei...
- Australia - Anne Jones activities June 2010..
- Philip Morris International to appeal Colombia's ...
- PAPER: Scotland Unhealthy risk factors those wi...
- Namibia - ready to implement the Tobacco Products ...
- Malaysia - best way to get teenage girls to stop ...
- Canada - farmers not given fair price for tobacco ...
- Brunei - smoking ban looks like it will work - EN...
- EU Court - two anti-smoking cases could lead to b...
- Singapore - new stricter tobacco legislation - sm...
- England - heart attack admissions fall after smoki...
- Australia - U.S. Chamber of Commerce expresses co...
- FDA CTR reports (2) on label descriptors and harm...
- Indonesia - on June 22nd will ask WTO to rule on U...
- U.S. - East Coast Cigarette Smuggling - on going..
- Armenia - conference results in future strategies ...
- Indonesia - PT Djarum Tobacco to sponsor badminton...
-
▼
06/13 - 06/20 (28)
© Copyright Notice: The content of this website is for information education purposes only and any newsbrief may be used only as "fair use" for information/education purposes with permission of the authors and providing that original references and associated reference links are included in HTML format.
1 comments:
March 8, 2013 at 5:01 AM
The Vapor King vapor cigarette successfully removes many of the downfalls typically associated with smoking Electronic Cigarettes Say goodbye to smokers breath, yellow teeth and the smell of smoke on your clothes. You can also get rid of all that smoking paraphernalia including lighters, ashtrays and cigarette butts.
Post a Comment